How Sleeping and Circadian Rhythms won the 2017 Nobel Prize in Physiology and Medicine
- Sep 28, 2018
- 3 min read
Ryan Bose-Roy
On October 4th, the Royal Swedish Academy of Sciences awarded the Nobel Prize in Physiology and Medicine to Jeffrey Hall, Michael Rosbash, and Michael Young for discovering the molecular mechanisms underlying circadian rhythms. These three scientists spent decades in their respective labs at the University of Maine, Brandeis University, and Rockefeller University using fruit flies to isolate the genes that control daily biological rhythms in humans.
We’ve known for years that humans have their own internal biological clock. This 24-hour cycle of hormones and chemicals allows us to sleep during the night and stay awake during the day, as well as to carry out a number of other processes necessary for maintaining bodily functions. When a mismatch occurs between our external environment and this internal clock - such as pulling an “all-nighter” or experiencing “jet lag” while traveling - the risk for various diseases markedly increases.
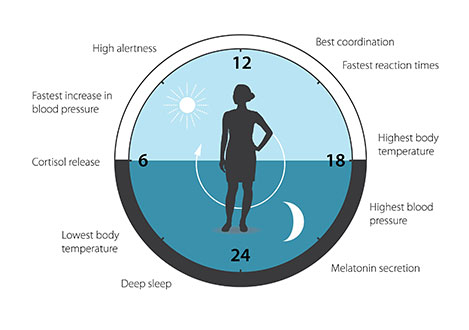
However, this “circadian rhythm” is not fixed; rather it constantly adapts to synchronize itself with the Earth’s revolutions. This is why our body eventually adapts to changes in time zones after traveling to faraway parts of the world.
Why does this happen? How are our bodies able to alter our biological rhythms so that wherever we are, we can always sleep at night and stay away during the day? These three researchers set out to find the answers.
In 1984, using fruit flies as model organisms, Jeffrey Hall and Michael Rosbash isolated a gene (called the “period gene”) that controls circadian rhythms by producing a protein that accumulates in cells during the night, and breaks down during the day. This protein, called PER, exhibits “negative feedback,” meaning that among other things, it actively blocks the gene that makes it. As more of this protein accumulates, it stops the production of the gene, causing its levels to drop after it can no longer be made. However, once the levels of PER fall, the gene that makes the protein becomes re-activated, and produces increasing amounts of PER, continuing the “circadian” cycle.
A few pieces of the puzzle were still missing, however. In order to block the activity of the period gene, PER would have to travel from the cytoplasm to the nucleus, where the gene was produced. Given that there was no existing mechanism for a molecule to enter the nucleus, the model was yet incomplete.
In 1994, Michael Young solved this problem by discovering a second clock gene. This gene, called “timeless,” produced a protein called TIM that, when bound to PER, allowed the resulting structure to enter the cell nucleus. Once there, the two proteins worked together to block the “period gene,” completing the negative feedback loop.

Nevertheless, there still remained the issue of why the circadian cycle operated in tandem with the 24-hour cycle. What controlled the frequency of these oscillations? Michael Young later identified another gene called “doubletime” which produced a protein termed DBT. This protein delayed the accumulation of the PER protein, slowing the cycle down.
As much else in the field of Science, a lot remains to be discovered. This year’s Nobel laureates elucidated the role of a protein that responds to light, identifying a mechanism by which light can synchronize our internal biological clocks, but the exact mechanism is still unclear.
Needless to say, research into the field of circadian rhythms is definitely something to stay awake for.




Comments